Hydrogel-Mediated Sensitivity Enhancement of Immunoassays
Most modern diagnostics, from ELISAs to microarrays, rely on capturing biomarkers on flat, two-dimensional surfaces. However, these 2D interfaces impose a physical ceiling on performance: they offer limited surface area for capture molecules, and the rigid environment can denature sensitive proteins or mask their active binding sites via unfavorable orientation. These constraints often lead to high background noise and restricted sensitivity, making it difficult to detect low-abundance biomarkers and low-affinity binders critical for early disease intervention.
We are overcoming these interfacial bottlenecks by transitioning from surface-bound models to a volumetric 3D framework. Our research utilizes highly hydrated, porous and functionalized PEG-based hydrogels that act as specialized scaffolds for diagnostic assays. By engineering the molecular architecture of these hydrogels, we enable a massive increase- over 3 orders of magnitude- in available capture sites compared to traditional 2D substrates. Using bio-orthogonal photo-click chemistry, our platform allows for the seamless integration of diverse capture probes, including antigens, antibodies and aptamers, with precise temporal and spatial control.
Using these versatile hydrogels, we are establishing a unified, 'plug-and-play' sensing interface- a standardized chemical architecture that yields significant sensitivity gains across a spectrum of readout modalities. This includes fluorescence and colorimetric readouts, electronic sensing on printed electrodes, and lateral flow microarrays. By bridging the gap between material science and device physics, our goal is to transform these high-performance 3D scaffolds into accessible, field-deployable tools for global health monitoring.
Next-Generation Lateral Flow Assays for Tuberculosis Diagnostics
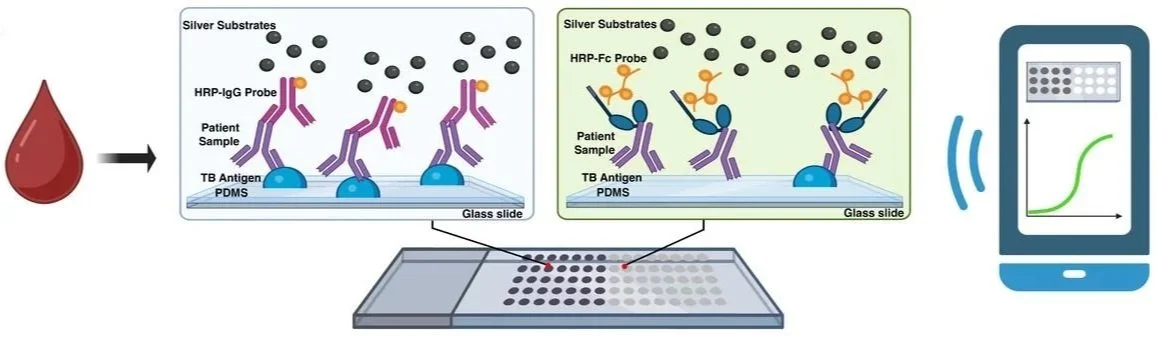
Tuberculosis (TB) remains the leading cause of death from a single infectious agent, yet nearly 40% of individuals who develop the disease go undiagnosed due to a lack of accurate and affordable point-of-care (POC) tools. Traditional serological tests typically rely on measuring antibody quantity (titer), which often fails to distinguish between active disease and latent infection. Our lab has identified antibody "quality"- specifically Fc functional properties like receptor binding- as a far more deterministic biomarker for identifying active TB in non-sputum samples.
We are working towards translating these sophisticated biomarkers into a simple, paper-based Lateral Flow Assay (LFA) format that prioritizes accessibility and usability in low-resource settings. A primary engineering challenge is that these target biomarkers are often present at low abundances and exhibit low binding affinities, making them difficult to detect with standard assays. To overcome these sensitivity limitations of traditional LFAs, we are developing LFA platforms featuring enhanced readouts, including fluorescence and enzymatic silver metallization. We are also exploring the integration of functionalized hydrogels within the nitrocellulose membrane to re-engineer local fluid dynamics and enable reliable, uniform multiplexability across high-density arrays. By bridging the gap between high-performance laboratory assays and field-deployable hardware, our goal is to provide a robust POC solution for TB and other high-sensitivity targets, such as cytokines.
Engineered Diagnostic Microdevices
Standard high-performance diagnostics like ELISAs are fundamentally limited by their reliance on expensive, bulky hardware and large sample volumes, creating a critical gap in resource-limited settings. To reach these communities, diagnostic technology must evolve into an inexpensive, portable, and sample-sparing format that maintains laboratory-grade precision outside of specialized infrastructure.
Our research engineers a suite of "lab-on-a-chip" platforms that replace centralized equipment with reconfigurable microarrays and signal amplification via enzymatic silver metallization for optical and electronic readouts. At the fundamental level, we are enhancing electronic biosensing by re-engineering the interfacial environment of interdigitated electrode (IDE) arrays. By integrating nanostructured surfaces- including gold nanowires and anodic aluminum oxide- with controlled silver metallization, we convert trace molecular binding events into massive, measurable changes in electrical impedance. We scale this innovation for high-content discovery through our µMAP platform, which utilizes reconfigurable microchannels to perform 1,400 simultaneous assays from just 15 µL of sample, enabling deep profiling of antibody "quality" with 1,000x less volume than traditional methods. Finally, we translate these principles into integrated, field-deployable hardware for COVID and TB diagnostics. These platforms utilize smartphone-interfaced readers to provide quantitative digital results for antibody titers and Fc functional features, bridging the gap between benchtop analysis and point-of-care delivery.
By bridging material science, microfluidics, and digital electronics, our goal is to deliver laboratory precision in the palm of your hand. We are building a future where high-content diagnostic data is accessible in any setting worldwide, transforming how we monitor our health.
Aptamer-Based Electronic Sensing for Continuous Monitoring
Conventional hormone testing relies on invasive blood draws and intermittent snapshots, often missing the critical cyclical variations inherent to endocrine health. To address this, we are developing a multiplexed, continuous biosensing platform capable of tracking real-time fluctuations in essential hormones- including estrogen, progesterone, LH, FSH, cortisol, and testosterone. This transition from static measurements to continuous data streams is vital for the accurate diagnosis and personalized management of complex endocrine-related conditions.
Our innovation centers on Electrochemical Aptamer-Based (EAB) biosensors, which provide a reagent-free alternative to traditional antibody-based assays. Unlike antibodies, aptamers exhibit fast, reversible binding kinetics and high thermal stability, enabling the tracking of concentration changes over time without external washes. We utilize structure-switching DNA aptamers immobilized on gold electrodes that elicit an electronic response post analyte binding. To maintain accuracy in physiological environments, we are integrating these sensors with specialized hydrogel networks designed to increase immobilization density, prevent biofouling, reduce signal drift, and hence maximize detection sensitivity.
Ultimately, we aim to translate this technology into wearable hormone monitors that empower both patients and clinicians with high-resolution, longitudinal data- shifting the diagnostic paradigm from isolated lab tests to real-time pattern analysis. Our goal is to provide a comprehensive window into human health, allowing for earlier detection of endocrine dysfunction and more precise medical interventions.